Article Text
Abstract
Introduction Healthy dietary choices have an important role in preventing chronic diseases such as cardiovascular disease (CVD). Increasing evidence suggests micronutrient intake (essential minerals and vitamins) is associated with abdominal aortic calcification (AAC), which is an advanced marker of CVD. However, the existing reports seem inconsistent. Some studies reported micronutrients are associated with a lower risk of AAC, while others have reported an increased risk. Therefore, this systematic review and meta-analysis sought to summarise the available evidence on the association of dietary micronutrient intake on AAC.
Methods and analysis A comprehensive systematic search of the PubMed/MEDLINE, EMBASE, Web of Science and Google Scholar databases from their inception up to September 1, 2024, will be conducted. All clinical studies that report eligible exposure/s (dietary micronutrient intake) and outcome/s (presence/severity of AAC) will be included, and this systematic review and meta-analysis protocol will be reported following the revised Preferred Reporting Items for Systematic Reviews and Meta-Analysis Protocols guidelines. The risk of bias for observational studies will be assessed using the Newcastle-Ottawa Scale and publication bias will be evaluated through visual inspection of funnel plots and the Egger’s and Begg’s regression tests. The Der Simonian and Laird random-effects model meta-analysis will be calculated to provide pooled results, and the weighted risk ratio with their 95% confidence intervals will be presented.
Ethics and dissemination The results will be disseminated through publishing in a peer-reviewed journal and public presentations at relevant local, national and international conferences, workshops and symposiums. Ethical approval is not required as this is a systematic review of publicly available data.
PROSPERO registration number
CRD42024575902
- Cardiovascular Disease
- NUTRITION & DIETETICS
- Drug Therapy
- Nutritional support
- GERIATRIC MEDICINE
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.
Statistics from Altmetric.com
STRENGTHS AND LIMITATIONS OF THIS STUDY
Sensitivity analysis will be carried out to assess the impact of the different dietary assessment methods, such as 24-hour recall, food frequency questionnaires and dietary records.
The main limitation of this review is the discrepancies in imaging modality, measurement and reporting of abdominal aortic calcification across studies, but we attempted to overcome this by exploring these aspects in prespecified subanalyses.
Further limitation is that this study has language restriction.
Introduction
Abdominal aortic calcification (AAC) is an advanced marker of cardiovascular disease (CVD), occurring in one out of every three individuals between the ages of 45 and 54, and nine out of 10 individuals aged 75 years and older.1 AAC is characterised by deposition of calcium within the arterial wall of the abdominal aorta.2 It is independently associated with not only CVD risk3 but also with muscle strength decline,4 lower bone mineral density, fractures,5 falls6 and dementia.7
Despite AAC’s association with debilitating medical conditions, the evidence regarding its prevention and available treatment is very limited. Randomised trials and cohort studies have examined the effect of intensive glycaemic treatment, vitamin B, calcium supplementation, phosphate binders and etidronic acid on AAC; however, the results show no influence on AAC occurrence or severity.8–15
Dietary strategies, particularly micronutrients such as potassium, zinc, selenium, magnesium, vitamins C and K, have gained significant attention for their pivotal role in the prevention of CVDs.16 However, the relationship between dietary micronutrient intake and AAC remains uncertain.17 One cohort study indicated that a higher intake of dietary zinc was associated with an 8% lower risk of severe AAC after adjustment for age, gender and ethnicity, though no association was found in the fully adjusted model.18 Similarly, higher consumption of dietary potassium,19 selenium20 and copper21 has been linked to a decrease in AAC incidence.20 Furthermore, dietary copper intake has been associated with a decreased risk of severe AAC.22
The relationship between dietary calcium intake and AAC is also complex. While higher dietary calcium intake has been associated with a lower prevalence of AAC at baseline, it has not been related to changes in the prevalence of AAC over 2 to 5 years.12 This finding is inconsistent with a cross-sectional study on diabetic mellitus patients, which found no significant association between dietary calcium and AAC progression.23 However, in healthy adults, there is an independent correlation between increased dietary calcium intake and severe AAC.24
A cross-sectional study in healthy participants found that increasing self-reported total magnesium intake by 50 mg/day resulted in a 12% decrease in the presence of AAC,25 and another study reported that a 0.1 mmol/L increase in serum magnesium (Mg) was independently associated with a 1.1 point decrease in AAC score.26
Higher dietary vitamin C intake has been linked to a reduced AAC score16 27 and a lower risk of AAC in adults.16 However, population-based studies have failed to demonstrate a significant correlation between dietary vitamin C intake and risk of severe AAC.27 Furthermore, there is a significant inverse relationship between higher folate (vitamin B9) intake and the risk of severe AAC,28 as well as a lower risk of AAC with higher dietary Vitamin A.29
Given the relationship between AAC and CVD, as well as the challenges it poses in terms of treatment, dietary micronutrient intake has drawn substantial attention for its potential role in preventing and ameliorating AAC.29 Although there are reasonable biological mechanisms that connect certain micronutrients to AAC, the current evidence is not conclusive. Additionally, the long-term effects of micronutrient intake on the presence/severity of AAC remain unclear.
Therefore, there is a need to conduct a systematic review and meta-analysis to synthesise the robust evidence regarding the effect of micronutrient intake on the development, severity or progression of AAC. Moreover, the association between specific dietary micronutrient intake and the presence and severity of AAC will be examined.
Methods and analysis
Eligibility criteria
Population
Adults with or at risk of AAC will be the population of interest.
Exposure
Micronutrient intake
The intervention is the intake of micronutrients such as vitamins A, B complex (B1, B2, B3, B5, B6, B7, B9, B12), C, D, E and K and calcium, iron, magnesium, zinc, selenium, iodine, copper, manganese and chromium. To measure the intake and status of the listed micronutrients, a combination of dietary assessment, biochemical tests and functional evaluations is typically used.
Comparator
For this systematic review and meta-analysis protocol, the reference group comprises individuals with lower intake or absence of the dietary micronutrients.
Outcome of interest
Primary outcome
The primary outcome will be the presence and severity of AAC. The presence of AAC identified from either radiography, dual X-ray absorptiometry (DXA) machine, MRI or CT scan and will be presented as AAC present or absent. Severity AAC will be categorised as low (lowest reported category), moderate (middle reported categories) and high (highest reported category).
Secondary outcome of interest will be the effect of specific dietary micronutrient intake in the severity and progression of AAC (rate of change in calcification volume, area or score over time). This review defines AAC as the presence of calcified deposits in the abdominal aorta identified from radiography, DXA, MRI or CT scan.
Setting and languages
This study will not restrict studies by settings, and only articles reported in English language will be included.
Study design
Observational cohort studies (both retrospective and prospective cohort studies), case–control studies and cross-sectional studies that report eligible exposure(s) and outcome(s) will be included in this review.
Inclusion criteria
Observational studies in humans. These include cohort (both retrospective and prospective cohort studies), case–control and cross-sectional studies that report eligible exposure(s) and outcome(s).
AAC assessed by any methodology.
Report of any dietary micronutrient effect on abdominal aortic outcome.
Exclusion criteria
We will exclude case series, case reports, commentaries, editor letters and reviews that do not contain any original data. We will exclude animal research from this review.
Search engines
To discover relevant studies, comprehensive literature searches on dietary micronutrient intake and AAC will be conducted in PubMed/MEDLINE, EMBASE, the Web of Science core collection, Google Scholar and CINAHL. Because no publication date limits were specified, the search will run from inception to September 1, 2024. The information sources will be updated prior to the submission of this evaluation. To ensure that all relevant studies are included in this review, the information sources will be augmented by examining the reference lists of selected studies and systematic reviews of comparable scope.
Search strategies
The search strategy for PubMed will be developed using a combination of medical subject headings (MeSH), keywords (text word) and [Title/Abstract] related to both exposure and the outcome of interest and combined using Boolean operators (AND, OR, NOT). The literature search will be conducted without any date constraints. The search strategies for PubMed/MEDLINE are provided in table 1. In addition to the MeSH terms, this systematic review and meta-analysis protocol will incorporate the PICO framework (Population, Intervention, Comparator, and Outcome).
Pilot search strategies in PubMed/MEDLINE database
We will apply the PICO framework in our review protocol as follows: Population: This targets individuals at risk of or diagnosed with AAC; Intervention: The intervention is dietary intake of micronutrients, mainly vitamins and minerals. Comparator: We will compare individuals with lower intake or absence of the dietary micronutrients. Outcome: Primary outcomes of interest will include presence/severity/progression of AAC, measured via imaging (eg, CT scans, lateral spine radiographs or echocardiography), while other reported outcome events can be considered secondary outcomes.
Protocol
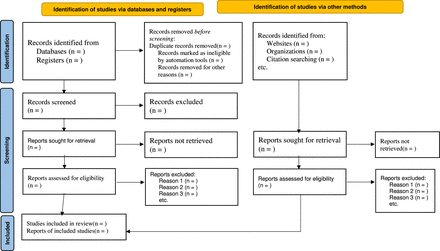
The systematic review and meta-analysis will be reported in accordance with the revised Preferred Reporting Items for Systematic Reviews and Meta-Analysis Protocols recommendations (figure 1). If protocol amendments are required after registration, the date, justification and details of the changes for each part will be provided.
Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) 2020 statement.32
Data management
Covidence will import, screen, store and analyse the results of the literature search. Covidence will automatically remove duplicates while also manually checking for study similarities (year of publication, author’s name, volume, issue, etc.) via the authors.
Selection of studies
EWT, ZHG, NAH and MBM will review the retrieved citations to determine study eligibility. In brief, the process for selecting studies for inclusion in the review and meta-analysis will be as follows: merge all identified records using EndNote, remove duplicate records of the same report, retrieve the full text of potentially relevant reports, link together multiple reports of the same study (using the first or largest report as the primary record and subsequent reports to supplement other data), examine full-text reports for compliance with eligibility criteria, correspond with investigators, where appropriate, to clarify study eligibility and request missing data and make final decisions on study inclusion.
Extraction of data
A data extraction excel form will be developed as a standard data collection instrument. For standardisation of the data extraction tool, a pilot test of the form will be done among five selected articles, and the form will be changed as needed based on group feedback. Furthermore, reviewers will conduct a calibration exercise to ensure that their evaluation methodologies are consistent across reviewers. And data from included studies will be extracted independently by three reviewers (EWT, NAH and MBM). In the event of a disagreement between the reviewers, the third investigator (ZHG) will be consulted.
Data items
The following data elements will be collected from the included studies: (i) study information: title, author name, year of publication, country of study, journal, sample size, study duration, study design, follow-up period and limitations. (ii) Population: characteristics of the participants, such as mean age, socioeconomic status, race or ethnicity and whether there was a diagnosis of AAC. (iii) Exposure: dietary micronutrient type and quantity. (iv) Outcome: a composite of outcome occurrences, the commencement of the follow-up period and the length of follow-up for outcome variables; (v) effect measures: reported effect measures for composite and distinct outcomes, if available, including p-values, SD and confidence intervals; and (vi) funding sources.
Risk of bias and quality assessment
The risk of bias for observational studies will be assessed using the Newcastle-Ottawa Scale. In addition, publication bias will be evaluated through visual inspection of funnel plots and the Egger’s and Begg’s regression tests. Summary estimates of confidence in the evidence will be assessed using the Grading of Recommendations Assessment Development and Evaluation (GRADE) of prognostic evidence. GRADE for evidence about prognosis starts with high-quality evidence that can then be rated down. These criteria are based on: (1) five domains diminishing confidence (−1 for risk of bias, inconsistency, imprecision, indirectness and publication bias) and (2) two situations increasing confidence (+1 or +2 for large–very large effect size and a +1 for a dose–response gradient). Two reviewers (NAH and MBM) will be assigned to evaluate the quality of each study, with a third reviewer (ZHG) consulted when conflicts emerge. The findings of the risk of bias assessment will be given in a table.
Statistical analysis and data synthesis
The analysis will be conducted using the R statistical software version 4.4.3.30
The analysis of outcome variables will be given according to either (1) AAC present or absent (2) AAC classified as 'low' (referent—lowest reported group) against 'high' (all other groups) or (3) dose–response where AAC was assessed in three or more groups.
We will use subgroup analysis to investigate clinical heterogeneity (general population, sex, race/ethnicity and age of cohort <60, 60–69, and ≥ 70 years old) and methodological heterogeneity (risk of bias of studies, imaging modality, such as radiograph, DXA or CT, and duration of follow-up for outcome measurement <5, 5–9 and ≥10 years). Meta-regression will be conducted using a random-effects model in the above subgroup categories.
Between study heterogeneity will be calculated using the I2 statistic with thresholds of 0–25%, 25%–49%, 50%–75% and above 75% indicating low, moderate, high and very high heterogeneity, respectively,31 where AAC events were reported individually, pooled risk difference and risk ratios with 95% confidence intervals will be generated, followed by a summary estimate using Der Simonian-Laird random-effects models. Furthermore, meta-analysis will be performed if the included studies are homogeneous to calculate the effect of dietary micronutrient intake on AAC.
Random effects will be used to determine the weights for the meta-analysis if necessary. A qualitative synthesis will be performed if there is significant heterogeneity (I² ≥ 50% or p<0.1) or in situations where the data are incomplete or unsuitable for meta-analysis. Moreover, when appropriate, additional analytical strategies will be evaluated to strengthen the robustness and depth of the analysis, such as subgroup analysis, sensitivity analysis or meta-regression. Furthermore, to minimise the risk of bias and ensure the reliability of the findings, studies with certain designs (eg, high risk of bias or methodological limitations) will be excluded from the analysis.
If various types of effect measures are utilised in the original studies, such as ORs, risk ratios and hazard ratios, the meta-analysis will be further conducted separately for each type of effect measure using R software. The study results will be reported in a sequential manner, starting with the primary outcomes, followed by secondary outcomes and important subgroup analysis. Based on design, setup and group classification methods, analysis will be conducted to investigate the possible causes of variability between studies and to explore the strength of the meta-analysis.
Meta-bias(es)
Outcome reporting biases will be assessed by comparing outcomes documented in research protocols with those reported in the actual study reports. Additionally, sensitivity analysis will be used to evaluate the impact of selective reporting on the results of meta-analyses, if deemed necessary. Funnel plots will also be employed to investigate potential publication bias. Furthermore, sensitivity analysis will be carried out to assess the impact of the different dietary assessment methods, such as 24- hour recall, food frequency questionnaires and dietary records.
Ethics and dissemination
The results will be disseminated through publishing in a peer-reviewed journal and public presentations at relevant local, national and international conferences, workshops and symposiums. Ethical approval is not required as this is a systematic review of publicly available data.
Ethics statements
Patient consent for publication
References
Footnotes
X @HagosZenawi
Contributors All the authors have made substantial intellectual contributions to the development of the protocol: EWT, NAH, MBM and ZHG contributed to the study concept and design. NAH and MBM formulated the search strategy. EWT led the writing of the manuscript and all authors conceived the conceptual ideas presented in the revised protocol critically. EWT and NAH drafted the risk of bias assessment section. ZHG developed the meta-bias and confidence in cumulative evidence sections. All authors read and approved the revised version and final supported versions. EWT has the primary responsibility for the final content. Etsay Weldekidan Tsegay is the guarantor.
Funding The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests None declared.
Patient and public involvement Patients and/or the public were not involved in the design, conduct, reporting or dissemination plans of this research.
Provenance and peer review Not commissioned; externally peer reviewed.