Article Text
Abstract
Objectives This study sought to estimate the effect of dexmedetomidine (DEX) administration on mortality in critically ill patients with acute kidney injury (AKI).
Design A retrospective cohort study.
Setting The study sourced its data from the Multiparameter Intelligent Monitoring in Intensive Care Database IV (MIMIC-IV), a comprehensive database of intensive care unit patients.
Participants A total of 15 754 critically ill patients with AKI were enrolled from the MIMIC-IV database.
Primary and secondary outcome Primary outcome was in-hospital mortality and secondary outcome was 180-day mortality.
Results 15 754 critically ill AKI patients were included in our analysis. We found that DEX use decreased in-hospital mortality risk by 38% (HR 0.62, 95% CI 0.55 to 0.70) and 180-day mortality risk by 23% (HR 0.77, 95% CI 0.69 to 0.85). After adjusting for confounding factors, DEX can reduce all three stages of AKI in in-hospital mortality.
Conclusions Our retrospective cohort study suggests that DEX significantly correlates with decreased risk-adjusted in-hospital and 180-day mortality in critically ill AKI patients. Nonetheless, future randomised controlled trials are warranted to validate our findings.
- intensive & critical care
- acute renal failure
- adult intensive & critical care
Data availability statement
Data are available on reasonable request.
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.
Statistics from Altmetric.com
STRENGTHS AND LIMITATIONS OF THIS STUDY
This study included 15 754 patients, which is a very large sample size for a clinical study of critically ill patients with acute kidney injury.
Additional confounding factors were adjusted and increased the reliability of our results and conducted a subgroup analysis of the association between dexmedetomidine use and in-hospital mortality.
This retrospective study was unable to conduct long-term follow-up, so we look forward to future multicentre clinical studies to make up for this deficiency and further verify the stability of the results in this study.
The data of this study were from a Multiparameter Intelligent Monitoring in Intensive Care Database IV database, and some data may be missing which slightly offsets the results.
Introduction
Over the past few decades, the prevalence of acute kidney injury (AKI) has significantly increased and has gradually become a global health concern.1 AKI is a common comorbidity among severely ill patients that require intensive care. Indeed, more than half of patients experience AKI during their stay in the intensive care unit (ICU).2 AKI is related to adverse outcomes, increases chronic kidney diseases (CKD) occurrence and kidney replacement therapy, and raises the risk of short-term and long-term deaths,3 causing huge social and economic burdens to patients and society. However, further research is warranted to optimise the management of AKI.4
In the context of critical care, effective sedation is of utmost importance for managing agitation and anxiety in patients.5 The primary objective of sedation in the ICU is to achieve a state of calmness and cooperation in the patient, allowing for the easy awakening and clear communication of needs, particularly concerning pain management.6 There is evidence to suggest that maintaining light sedation in patients in the ICU can lead to better outcomes. Current guidelines recommend dexmedetomidine (DEX) for sedation in an intensive care setting.7 DEX is widely used in ICU as a highly selective alpha-2 agonist.8 An increasing body of evidence suggests that DEX can inhibit the production of excess inflammation cytokine and protect renal function, which may positively impact the prognosis of AKI.9–11 However, the renoprotective effects of DEX in critically ill patients have not been explored, based on the above characteristics of DEX, we assume that the use of DEX can reduce the mortality rate of AKI patients. The current literature consists mainly of basic-level studies or small samples of other population cohorts, with a paucity of large-scale research.12 13 Hence, the present study aims to investigate DEX’s effect on AKI-related mortality in critically ill patients based on a large sample of critical care public databases.
Methods
Data sources
The study sourced its data from the Multiparameter Intelligent Monitoring in Intensive Care Database IV (MIMIC-IV), a comprehensive database of ICU patients.14 We collected data on AKI patients from the MIMIC-IV database treated with or without DEX. This database contains a publicly available and real-world clinical database of patients at the Beth Israel Deaconess Medical Center from 2008 to 2019. Informed consent of patients was not required in this study since confidential patient information was already deleted. All reports followed the guidelines of Strengthening Epidemiological Observation and Research Report.15 A Collaborative Institutional Training Initiative licence (Certificate No. 11326088) was obtained by WW, who was entitled to extract data from the MIMIC-IV database in accordance with the relevant regulations.
Participants
This study included patients who were admitted to the ICU and diagnosed with AKI according to the Kidney Disease Improving Global Outcomes criteria.16 The definition of baseline serum creatinine level (SCr) in this study was based on two criteria: (1) the minimum SCr level recorded within 7 days before ICU admission or (2) if there were no SCr data available before admission, the first SCr level measured on admission to the ICU was used as the baseline.17 The MIMIC IV 2.0 database only contains data on adults older than 18. Patients who met any of the following criteria were excluded from the study: (1) death within 48 hours after admission to the ICU and (2) ICU stays less than 48 hours.
Covariates
The study included demographic characteristics and clinical characteristics with 24-hour average values. The Sequential Organ Failure Assessment (SOFA) score,18 and Simplified Acute Physiology Score (SAPS) II,19 were calculated within the first 24 hours after the ICU admission. We collected the following clinical information of each patient: demographics, laboratory measurements, vital signs, ethnicity, admission type, need of support, comorbidities at ICU admission, severity of illness, sedative-analgesic medications use, AKI stage, sepsis, antibiotic use, glucocorticoid use(table 1). These covariates, including clinical characteristics and basic demographic information, were based on other relevant studies and clinical practice.20–22
Baseline characteristics for the two groups before and after matching
Data definitions
Extracted data from MIMIC-IV on the first day of ICU admission, including age, gender, laboratory measurements, vital signs, ethnicity, admission type, vasopressors, mechanical ventilation (MV), continuous renal replacement therapy (CRRT), comorbidities, SOFA score, SAPS II score. We also collected information on whether DEX, propofol, midazolam, fentanyl, antibiotics and glucocorticoids were used during ICU hospitalisation. Sepsis was defined as a life-threatening organ dysfunction caused by a dysregulation of host response to infection (Sepsis 3.0),23 which refers to patients with documented or suspected infections and acute changes in the SOFA score≥2 points.
DEX use was defined as patients who received any DEX treatment throughout hospitalisation in the ICU.
Outcomes
In-hospital mortality refers to the death of patients during this hospitalisation, which was recorded by the hospital.24 In the MIMIC-IV database, this type of patients will be marked with a ‘hospital expire flag’ to indicate the hospital death during this hospitalisation. If a patient has been hospitalised multiple times, we only select the first check-in record. The primary outcome was in-hospital mortality and the second outcome was 180-day mortality of AKI patients in the ICU.
Statistical analysis
Our study presented continuous variables described as mean±SD for normally distributed or as the median and IQR if not normally distributed, while the t-test or Mann-Whitney U test was used for comparison between groups, respectively. Categorical variables were presented using numbers and percentages (%), and the χ2 test or Fisher’s exact test was adopted from group comparisons. We used propensity score matching (PSM) by 1:1 nearest neighbour matching to adjust the baseline difference between the groups. The calliper value was set to 0.2 between matching participants. The standardised mean difference (SMD) was calculated to determine the balance within the model (table 1), and SMD greater than 0.1 was considered unbalanced.25
We used Cox proportional hazards regression and binary logistic regression to assess the effect of DEX use on in-hospital mortality and 180-day mortality. In the subgroup analysis, we used binary logistic regression analysis of in-hospital mortality to assess the effect of DEX use on in-hospital mortality in subgroup populations. Parameters with a p<0.1 during univariate analysis and potential confounding factors were included in the multivariate regression model. In the subgroup analysis, we classified patients based on age, gender, AKI stage and sepsis incidence to assess the reliability of our findings. We conducted all statistical analyses using either R V.4.1.2, GraphPad Prism V.6 software, or MedCalc V.20.1. The significance level was set at p<0.05.
Patient and public involvement
Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
Results
Baseline population characteristics
A total of 15 754 individuals diagnosed with AKI after ICU admission within 48 hours were selected for this analysis after excluding patients based on the exclusion criteria (figure 1). In the whole cohort, the median age of patients was 68 years old, with 8998 (57.1%) males and 6756 (42.9%) females. A toal of 8548 (54.3%) patients were diagnosed with AKI stage 1, 5695 (36.1%) with AKI stage 2 and 1511 (9.6%) with AKI stage 3.
Flow chart of patients selection for the study. AKI, acute kidney injury; ICU, intensive care unit; MIMIC-IV, Medical Information Mart for Intensive Care Database IV.
In this study, patients were classified into non-DEX (n=12 536) and DEX (n=3218) groups. Compared with the non-DEX group, the DEX group was younger, heavier weight, higher white cell count, temperature, peripheral oxygen saturation (SpO2), SOFA score and SAPS II score, faster respiratory rate and heart rate, lower platelet, creatinine, bun, lactate, congestive heart failure and renal disease, more female, white ethnicity, emergency admission, vasopressin use, MV, CRRT, chronic pulmonary, liver disease, propofol use, midazolam use fentanyl use AKI stage 1, sepsis and antibiotics use. After PSM, the characteristics of 3196 patients in both groups were comparable (SMD<0.1) (table 1).
Primary outcome
In-hospital mortality: We found that DEX use decreased in-hospital mortality risk by 38% among critically ill patients with AKI (HR 0.62, 95% CI 0.55 to 0.70) during PSM modelling. Moreover, Cox proportional hazards regression (HR 0.61, 95% CI 0.55 to 0.68) (figure 2, online supplemental table 1) and binary logistic regression (OR 0.64, 95% CI 0.56 to 0.73) (online supplemental table 2) were consistent with the trend of PSM modelling. A significant decrease in in-hospital mortality was observed among critically ill patients receiving DEX at AKI stage 1 (15.6% vs 10.7%, p<0.001), stage 2 (18.5% vs 14.7%, p=0.017) but not stage 3 (27.6% vs 26.6%, p=0.848) (figure 3).
Supplemental material
Association between in-hospital mortality, 180-day mortality and dexmedetomidine use of AKI patients evaluated by the Cox model. Unadjusted: without adjustment; multivarable adjusted: adjusted for all the baseline variables shown in table 1. AKI, acute kidney injury; PSM, propensity score matching.
In-hospital mortality and 180-day mortality of AKI in critically ill patients between the dexmedetomidine and non-dexmedetomidine group in different AKI stage. AKI, acute kidney injury. *p< 0.05 between the two groups.
Second outcome
A 180-day mortality: DEX use also reduced 180-day mortality risk by 23% among critically ill patients with AKI (HR 0.77, 95% CI 0.69 to 0.85) during PSM modelling. Moreover, Cox proportional hazards regression (HR 0.68, 95% CI 0.63 to 0.74) (figure 2, online supplemental table 3) and binary logistic regression (OR 0.64, 95% CI 0.57 to 0.71) (online supplemental table 4) were consistent with the trend of PSM modelling. A significant decrease in 180-day mortality was observed among critically ill patients receiving DEX at AKI stage 1 (24.7% vs 18.2%, p<0.001), stage 2 (28.3% vs 24.0%, p=0.023) but not stage 3 (39.1% vs 38.3%, p=0.861) (figure 3).
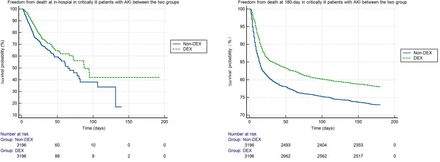
We conducted a check on the goodness of fit of the model and found that p<0.05, indicates a good fit of the model. We also investigated the in-hospital and 180-day survival in critical patients with AKI. The DEX group had significantly higher freedom from death at in-hospital (86.5% vs 82.4%, p<0.001) and 180-day (78.0% vs 72.8%, p<0.001) than the non-DEX group, respectively (figure 4).
Freedom from death at in-hospital and 180-day in critically ill patients with AKI between the two groups. AKI, acute kidney injury; DEX, dexmedetomidine.
Overall, the use of DEX had a significant beneficial effect on the in-hospital mortality and 180-day mortality of AKI in critically ill patients.
Subgroup analysis
Subgroup analysis indicated that DEX use reduced in-hospital mortality of critically ill patients with AKI (online supplemental figure 1). There were no interactions between age, gender, sepsis, AKI stage and DEX use, suggesting that these results were comparable for all populations.
Discussion
The present study showed that DEX use in severely ill AKI patients was linked with lower risk-adjusted in-hospital mortality and 180-day mortality. Consistent results were observed in different models. During subgroup analysis, after stratification according to age, gender, AKI stage, and sepsis, a strong correlation was still observed. Overall, we provide preliminary evidence that DEX has a beneficial effect on the prognosis of AKI in critically ill patients, providing the foothold to improve the outcomes of this patient population.
Overwhelming literature substantiates that DEX can alleviate AKI caused by several factors. Ruegg et al in a review summarised the role of DEX in preventing AKI in intensive care.26 Wang et al previously uncovered that DEX could ameliorate AKI in mice with sepsis by partially inhibiting oxidative stress and apoptosis by modulating the p75NTR/p38MAPK/JNK signalling pathways.27 Zhao et al further substantiated that DEX protected against lipopolysaccharide-induced AKI by promoting autophagy mediated by PI3K/AKI/mTOR pathway inhibition.12 A meta-analysis by Loomba et al demonstrated that DEX could confer postoperative renal protective effects with lower NGAL levels and increased creatine clearance in patients who received DEX. These effects correlated with reduced ICU length of stay and risk of AKI and mortality.13 Shan et al found that DEX could minimise AKI incidence in Stanford type B aortic dissection patients after endovascular aortic repair.10 A single-centre randomised controlled trial (RCT) of 108 patients28 showed that prior administration of DEX within 24 hours after induction of anaesthesia could reduce the incidence of AKI after aortic surgery under cardiopulmonary bypass. An RCT by Tang et al29 showed that DEX pretreatment attenuated AKI in patients. Animal studies further indicated that DEX could reduce cellular injury, improve renal function and mitigate apoptosis in renal cells. Furthermore, Liu et al revealed that DEX infusion in ICU patients with septic shock was linked to decreased AKI incidence and reduced ICU stay and CRRT performance. It is highly conceivable that the mechanism is related to anti-inflammatory effects and immunomodulation.30
Inflammation is key to AKI pathogenesis, progression and prognosis. An increasing body of evidence suggests that DEX confers renoprotective effects and may be involved in the regulation of inflammation.31–33 A meta-analysis by Wang et al9 that included 4842 patients showed that DEX decreased proinflammatory like cytokines interleukin-6, C reactive protein, tumour necrosis factor-α and increased anti-inflammatory cytokines like IL-10 in surgical patients. A subanalysis of a multicentre RCT by Ohta et al34 suggested that sedation using DEX reduced inflammation in patients with sepsis requiring MV. Animal studies have shown that DEX may have a protective effect on cisplatin-induced AKI, and its potential mechanism may be related to the regulation of apoptosis and inflammatory response.35 In addition, DEX can ameliorate microcirculation disorders by decreasing norepinephrine levels in the blood and increasing urine output and renal blood flow.36 Since our study did not collect inflammatory indicators, the hypothesis that DEX may attenuate excessive inflammation could not be confirmed, emphasising the need for future studies. Emerging evidence substantiates the benefits of DEX in AKI, but the prognosis and follow-up of AKI in critically ill patients have been largely understudied. In the present study, we consistently found that the in-hospital mortality of AKI patients in the DEX group was significantly lower than in the non-DEX group. At the same time, we found that the 180-day mortality of AKI patients was consistent with the in-hospital mortality, suggesting that DEX use is associated with survival benefits in this particular patient population. Our research provides a theoretical basis for clinicians to use DEX to manage critically ill patients with AKI. In our research results, we also found that propofol reduced the risk of in-hospital mortality and 180-day mortality, and we speculate that this may be due to the renal protective effect of propofol,37 38 this finding may also be due to the fact that propofol may be give more often to patients who are haemodynamically more stable and therefore more like to survive. The commonly used sedative drugs in ICU are propofol, DEX and midazolam, which can be used alone or in combination. There was no statistically significant difference in the use of propofol between the two groups after PSM, and we also adjusted for propofol as a confounding factor in the logistic regression analysis, and the interference of propofol on death outcomes was excluded.
Our subgroup analysis showed that DEX was effective in sepsis-associated-AKI (SA-AKI) patients, consistent with the literature.21 Consistently, Hu et al analysed 2192 patients with SA-AKI and found that DEX use was related to decreased in-hospital mortality and improved renal function recovery of SA-AKI in critically ill patients. Unlike Hu’s study is that our study included all types of AKI populations in the ICU. Our results showed that DEX use reduced in-hospital mortality of AKI in critically ill patients. Follow-up analysis showed that DEX use reduced the 180-day mortality of patients. Our findings suggest DEX is effective against sepsis-associated AKI and for AKI patients in general and improves the long-term prognosis. The role of DEX on more types of AKI subgroups warrants further exploration in severely ill subjects. Our study found that using DEX in critically ill patients with AKI can reduce in-hospital mortality and 180-day mortality. In figure 3, we conducted a χ2 test from group comparisons and the impact of confounding factors was not included. In the subgroup analysis in online supplemental figures, we used binary logistic regression analysis and include the influence of confounding factors. After adjusting for confounding factors, DEX can reduce all three stages of AKI in in-hospital mortality. Shehabi et al39 found that early use of DEX sedation can reduce the 90-day mortality in elderly patients with critically ill MV in the sedation practice in intensive care evaluation (SPICE III) trial, whether the patient has surgery or not. On the contrary, a higher likelihood of an increase in 90-day mortality was observed in younger patients of non-surgical status. However, it has not been thoroughly researched for the use of DEX sedation in critically ill patients with AKI, and this study can serve as a supplement to such patients in the SPICE III trial.
Our research has several limitations. First, data acquired from this database was adopted to maximise generalisability and power. Accordingly, there was no formal calculation of sample size in this study. Although the sample size of the subgroup was comparatively larger compared with previous studies, it may also increase the risk of false positive results during multiple subgroup analyses. Second, our study’s retrospective nature may have limited our findings’ accuracy, and there could be other unknown potentially confounding factors that we were unable to control for. Third, we adjusted for many confounding factors, and PSM was conducted. Moreover, data analysed in this study were acquired from a single-centre observation database, emphasising the need for a multicentre RCT to increase the robustness of our findings. Forth, due to the lack of admission diagnosis recorded in the MIMIC database, it is difficult for us to accurately identify the aetiology of AKI in each patient. Therefore, the AKI patients defined in this study are actually unselected AKI. Although it is difficult to determine the exact cause of AKI and the reason why patients are admitted to the ICU, we have made necessary adjustments for other confounding factors that affect patient mortality. Our conclusion is stable and reliable, and may only apply to unselected AKI in critically ill patients. Fifth, this study did not consider the dosage and duration of DEX use, and further attention is needed in future studies. Sixth, in this study, we did not consider changes in exposure or covariates over time. Due to the large number and heterogeneity of patients, it is difficult to quantify or qualitatively measure the changes in exposure or covariates of all patients over time. We focus on the measurement indicators of patients at admission, and only by analysing this time point can we have significant value in promoting and applying our conclusions in clinical practice.
Conclusion
This retrospective cohort study showed that DEX administration is associated with reduced risk-adjusted in-hospital and 180-day mortality in critically ill patients with AKI. However, further RCTs are needed to develop the robustness of our findings.
Data availability statement
Data are available on reasonable request.
Ethics statements
Patient consent for publication
Acknowledgments
We would like to thank the participants, patient advisers, developers and investigators associated with the Medical Information Mart for Intensive Care (MIMIC)-IV database.
References
Supplementary materials
Supplementary Data
This web only file has been produced by the BMJ Publishing Group from an electronic file supplied by the author(s) and has not been edited for content.
Footnotes
Contributors WW and JL designed the study. YJ and PZ conducted data collection. PG and HW conducted data analysis. WW wrote the manuscript. WW and JL analysed and interpreted the result. JL was responsible for the overall content as the guarantor. All authors reviewed this manuscript.
Funding This work was supported by the National Natural Science Foundation of China (Grant Number: 81960669) and the National High Level Hospital Clinical Research Funding (Grant Number: 2022-GSP-GG-22).
Competing interests None declared.
Patient and public involvement Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
Provenance and peer review Not commissioned; externally peer reviewed.
Supplemental material This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.